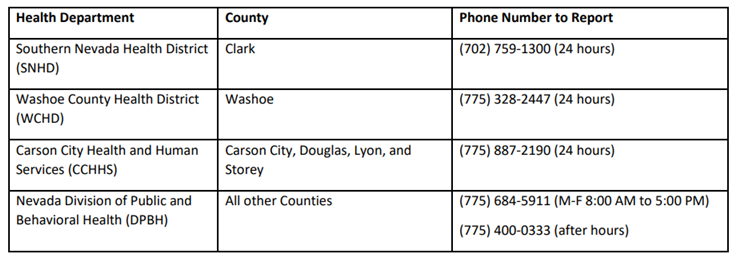
Guidance taken from CDC Laboratory Outreach Communication System advisory Laboratories with specimens that are suspected to contain monkeypox virus should contact their hospital infection control personnel (if part of a hospital system) or their state or local public health department (see contact list below). The Centers for Disease Control and Prevention (CDC) Emergency Operations Center (EOC) is also available for guidance at (770) 488-7100. After consultation, laboratories can send appropriately collected specimens for real-time polymerase chain reaction (PCR) testing to the Nevada State Public Health Laboratory (NSPHL) or the Southern Nevada Public Health Laboratory (SNPHL), unless authorized to send specimens directly to CDC. For more information, see the CDC Laboratory Process for Monkeypox Virus Testing. Clinical laboratories should follow these guidelines when collecting specimens for monkeypox virus: (Note: Consultation with the state health department and CDC should be performed prior to collecting specimens.)
Questions?
1 Comment
|
AuthorThe Bureau of Health Care Quality and Compliance (HCQC) licenses medical and other health facilities, child care facilities and personnel, and medical laboratories and personnel in Nevada. HCQC also conducts compliance surveys and takes complaints. Archives
October 2023
Categories
All
|
 RSS Feed
RSS Feed
