Training about this emerging fungus will cover prevention, transmission, testing and more A free webinar about Candida auris, an emerging and often multi-drug reistant fungus that presents a serious global health threat, will be held at 10 a.m. on June 28. The webinar is a joint effort of the Nevada Office of Public Health Investigations and Epidemiology (OPHIE), Washington State Department of Health and Centers for Disease Control and Prevention (CDC). Additional information about Candida auris can be found in the technical bulletin linked here.
The objectives of the webinar are:
The webinar has been approved for 1 nursing CEU. Questions can be emailed to [email protected].
0 Comments
Technical bulletin from state health officials provides clinical guidance The U.S. Food and Drug Administration (FDA) has authorized the emergency use of the Moderna COVID-19 vaccine and the Pfizer-BioNTech COVID-19 vaccine for the prevention of COVID-19 to include use in children under 5 years of age.
On June 18, the day after FDA authorization, the Advisory Committee on Immunization Practices (ACIP) unanimously voted to recommend the use of the Moderna COVID-19 vaccine two-dose primary series for children ages 6 months to 5 years and the Pfizer three-dose series for children ages 6 months to 4 years. According to disease burden data to the ACIP, children ages 6 months through 4 years are at risk of severe COVID -19 infection and more than 2 million cases of the illness have been identified among this population. COVID-19-associated hospitalizations among children ages 6 months through 4 years have similar severity compared to older children and adolescents and more than half of the hospitalized children ages 6 months through 4 years had no underlying conditions. The burden of COVID-19associated death among this population is similar to or exceeds that of other pediatric vaccine-preventable diseases. For more information, including vaccine administration guidance, see the technical bulletin on this topic posted to the Division of Public and Behavioral Health Technical Bulletins web page. Health facilities are encouraged to adopt concepts to be prepared for future emergencies Nevada’s Crisis Standards of Care (CSC) have been officially deactivated, effective June 10, 2022, however health facilities are encouraged to implement policies and procedures to be prepared for unforeseen circumstances. CSC deactivation has hinged upon the removal of Nevada’s emergency declaration and associated Emergency Directive 011, which officially was terminated on May 20, 2022. Additionally, since March 2022, health care systems have reported a shift from CSC levels to contingent, or conventional, standards of care. The State Chief Medical Officer, based on Emergency Providers of Nevada recommendations, and in consultation with the Department of Health and Human Services Director, the Governor’s Office, the Attorney General’s Office, local health officials, and Division of Emergency Management, is officially deactivating the Nevada CSC Plan. The department’s Bureau of Health Care Quality and Compliance (HCQC) encourages licensed health facilities to make the concepts discussed in CSC part of the facility’s own internal disaster plan. Although the declaration of emergency is no longer in effect, it doesn’t mean that facilities won’t experience internal disasters and the CSC are a way to ensure basic level services are maintained to mitigate the safety of patients/residents.
State health officials urge clinicians to take screening, treatment and prevention actions with patients In 2020, Nevada ranked first in the nation in reported cases of primary and secondary (P&S) syphilis and fourth in the nation for congenital syphilis (CS). While the rate of syphilis among males is much higher than among females, over the past several years there has been a rise in syphilis cases among women, both nationally and in Nevada.
The Nevada Department of Health and Human Services has issued a technical bulletin to inform clinicians and health authorities about:
Recorded webinar from Nevada OSHA discusses new requirement for medical facilities under NRS 618 A recorded webinar titled, “Violence at Nevada Medical Facilities Regulations,” that discusses Nevada regulations that took effect Dec. 22, 2021, is available for viewing on YouTube by affected facilities. The webinar was presented on March 16, 2022, by Michael Rodrigues of the Nevada Occupational Safety and Health Administration (NV OSHA).
Click here to view the video. Below is a summary of the regulations:
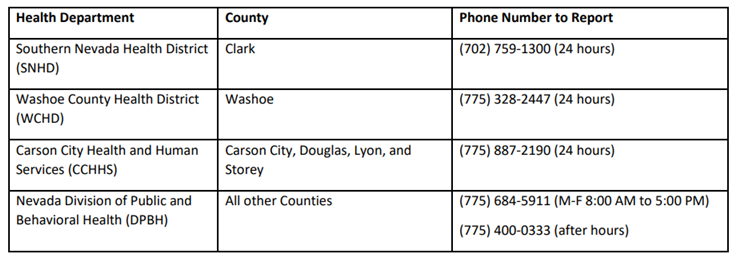
Read the full letter from OSHA about these regulations. For more information, call (702) 486-9061 or e-mail [email protected]. Guidance taken from CDC Laboratory Outreach Communication System advisory Laboratories with specimens that are suspected to contain monkeypox virus should contact their hospital infection control personnel (if part of a hospital system) or their state or local public health department (see contact list below). The Centers for Disease Control and Prevention (CDC) Emergency Operations Center (EOC) is also available for guidance at (770) 488-7100. After consultation, laboratories can send appropriately collected specimens for real-time polymerase chain reaction (PCR) testing to the Nevada State Public Health Laboratory (NSPHL) or the Southern Nevada Public Health Laboratory (SNPHL), unless authorized to send specimens directly to CDC. For more information, see the CDC Laboratory Process for Monkeypox Virus Testing. Clinical laboratories should follow these guidelines when collecting specimens for monkeypox virus: (Note: Consultation with the state health department and CDC should be performed prior to collecting specimens.)
Questions?
Health facility inspectors will begin citing deficiencies and assigning scope and severity Effective July 1, 2022, health facility inspectors will begin citing deficiencies and assigning scope and severity on health facilities found to be out of compliance with the state’s cultural competency requirements under Nevada Revised Statutes 449.101, 449.103 and 449.104.
Under these additions signed into Nevada law in 2019, all state-licensed medical facilities and facilities for the dependent are required to:
Note: The Bureau of Health Care Quality and Compliance would like to thank facilities that have already complied with these requirements. This collaborative effort will help to ensure a culturally competent health care system in Nevada with facilities that provide diverse and socially appropriate services to all patients and reduce health disparities. Doctors, nurses and other facility staff who have effective interpersonal and working relationships that supersede cultural differences are essential to successful health care outcomes for patients and residents. |
AuthorThe Bureau of Health Care Quality and Compliance (HCQC) licenses medical and other health facilities, child care facilities and personnel, and medical laboratories and personnel in Nevada. HCQC also conducts compliance surveys and takes complaints. Archives
October 2023
Categories
All
|
||||||
 RSS Feed
RSS Feed
