CDC revises guidance to include mixing and matching of initial and booster doses The Centers for Disease Control and Prevention (CDC) has issued new guidance that eligible individuals may choose which of the three COVID-19 vaccines available in the United States that they receive as a booster dose.
This week, the CDC’s Advisory Committee on Immunization Practices (ACIP) met to discuss the recent booster dose recommendations for both the Moderna and Janssen/J&J COVId-19 vaccines. Some people may prefer the vaccine type they originally received, while others may prefer to get a different booster. As a result of this week’s meetings the CDC’s recommendations now allow for this type of mix and match dosing for booster shots. For more information, see the Division of Public and Behavioral Health technical bulletins web page.
0 Comments
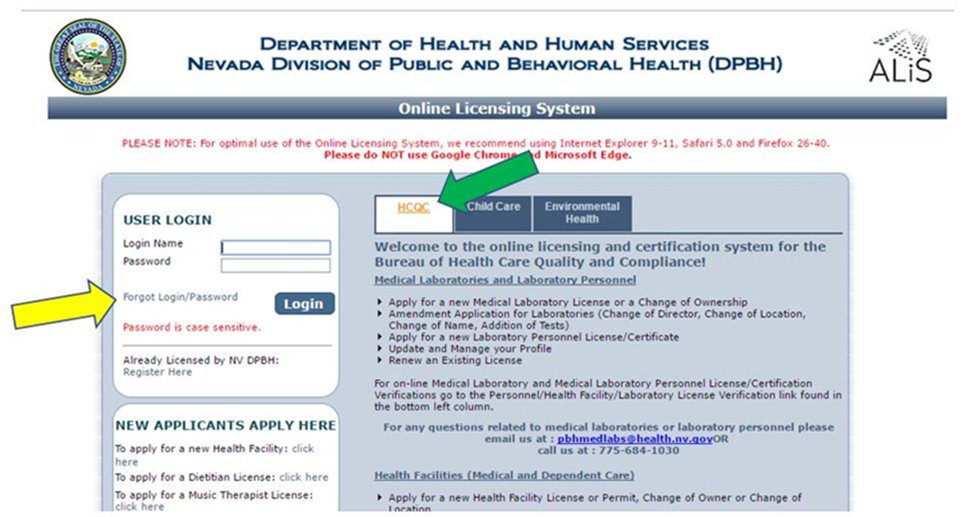
Deadline to renew is Nov. 15, so make sure your email is current in the online system Nevada health facilities can now submit their license renewal applications to take effect Jan. 1, 2022. Renewal applications must be submitted on or before Nov. 15, 2021.
Renewal notifications were sent on Oct. 1 to the email address in the Nevada online licensing system account for your facility. Be sure to check and update (if necessary) the facility’s contact email address in the Nevada online licensing system because those email addresses were used to send renewal notices and will be used by the Bureau of Health Care Quality and Compliance (HCQC) to further renewal notifications, reminders and other information. All of Nevada’s health facility licensing is done online, and the system can be accessed easily by typing myhealthfacilitylicense.nv.gov into your web browser address bar. Below are some helpful hints for renewing your health facility license:
Technical bulletin addresses issues related to point-of-care tests used to detect current COVID-19 viral infection. State health officials have issued updated guidance regarding Point of care (POC) COVID-19 antigen tests, which are one testing option available to respond to the COVID-19 pandemic. Antigen tests are immunoassays that detect the presence of a specific viral antigen, which implies current viral infection. Antigen tests are currently authorized to be performed on nasopharyngeal or nasal swab specimens. The U.S. Food and Drug Administration (FDA) has granted emergency use authorization (EUA) for antigen tests that can identify COVID-19.
The technical bulletin with updated guidance addresses a number of issues important to health facilities:
|
AuthorThe Bureau of Health Care Quality and Compliance (HCQC) licenses medical and other health facilities, child care facilities and personnel, and medical laboratories and personnel in Nevada. HCQC also conducts compliance surveys and takes complaints. Archives
October 2023
Categories
All
|
 RSS Feed
RSS Feed
